FDA
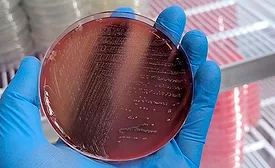
Tips for Building Confidence in Your Third-Party Laboratory's Salmonella Results
Salmonella testing plays an important role in reducing risk by providing validation and verification of Salmonella control strategies
Read More
FDA FY 2027 Budget Request Includes $57 Million for ‘MAHA’
While FDA's Human Foods Program gets increased funds, USDA-FSIS' budget stays stable.
April 7, 2026
Never miss the latest news and trends driving the food safety industry
Newsletters | Website | eMagazine
JOIN TODAY!Copyright ©2026. All Rights Reserved BNP Media, Inc. and BNP Media II, LLC.
Design, CMS, Hosting & Web Development :: ePublishing