Improvements in Hand Hygiene Using a Dry Paper Towel Containing Benzalkonium Chloride
Introduction
Hand hygiene continues to be the most effective means to prevent infection and its spread. Unfortunately, compliance with proper handwashing technique continues to be an issue in several sectors including health care[1] and foodservice.[2] According to the World Health Organization, proper handwashing involves the use of water, soap, lathering, rubbing, rinsing and drying preferably with a paper towel.[3] Compliance in these two sectors continues to be poor either in the practice of handwashing itself or in the various stages of the handwashing process. To improve the efficacy of hand hygiene, the use of hand sanitizers has been implemented in the health care, food service, and consumer environments. The presence of a sanitizer acts as a supplement to handwashing and is the best option in areas where water is unavailable. Yet even in the presence of hand sanitizers, there is a requirement for active cues whether visual[4] or observational.[5] When water, soap and drying towels are available, however, the use of handwashing is optimal when done properly.[6]
A paper towel that incorporates several of the needs for improved hand hygiene compliance has recently been developed by Cascades Tissue Group. The Cascades® Antibacterial paper towel product, AB, is a dry paper towel in which the antimicrobial agent, benzalkonium chloride (BC), has been included in the product. In addition to the incorporation of a sanitizer in the fiber, the towel is colored green to provide a more visually aesthetic cue to those undergoing the handwashing process. In the laboratory environment, third-party testing has shown thatthe towel has demonstrated the ability to remove >4 log of bacteria including methicillin resistant Staphylococcus aureus (MRSA), Escherichia coli, Salmonella and Listeria monocytogenes, among others (Dr. Ismail Fliss, Laval University; Kyle McGovern, Bioscience Laboratories; SM Laboratories). Moreover, there is a bacteriostatic effect that persists for up to two hours. Yet laboratory conditions are specific and highly structured, and may not reflect the activity of the product in the field.[7] Thus, while testing has shown that the product is effective, a field test was warranted.
To determine the efficacy of AB in the field, a study was conducted. The goal of the work was to determine whether the addition of this product in the handwashing process could improve the efficacy of handwashing in the field and reduce bacterial contamination on hands. The test involved the use of the fingertip touch method[6] to identify differences between the level of hand contamination prior to washing and immediately after washing. In addition, AB was compared against the standard issue white paper towel C in the field to determine any statistically significant differences.
Methods
Test subjects
The field test was performed on October 17, 2012 at the International Sanitary Supply Association conference in Chicago. A booth was set up at the conference and 14 volunteers accepted participation (7 for the test and 7 controls). They were given a set of instructions to follow and asked for a verbal agreement. Upon agreement, volunteers were escorted by a member of the trial team to the test area.
Materials and equipment
AB towels were provided by the company Cascades Tissue Group. All finger dish tests were performed using 100 mm petri dishes containing Trypticase soy agar (Situ Biosciences, Chicago). Plates were incubated for 24 hours at 98.6 ºF (37 ºC) in an NQ9 temperature controlled incubator (Darwin Chamber Company, St. Louis).
Test method
Volunteers were asked to first place the three fingers of their right hand on an open petri plate for a period of 30 seconds. The volunteers were asked to press firmly to ensure that an impression of the fingerprint would be left behind. After 30 seconds, the petri dish was labeled with i) the initials of the volunteer; ii) the letters B4 for before; and iii) either C or AB for designation of normal paper towel or antibacterial towel, respectively.
Based on the field test the subjects were given either the AB towel or the C towel (for conference towel).Volunteers chosen to use the AB towel were escorted to a nearby washroom (less than 50 feet from the test area) and provided with two pieces of the AB towel. Volunteers chosen to use the C towel were escorted to the washroom and asked to dry their hands with the normal white paper towel provided in the public restroom. Additionally, volunteers were asked to wash their hands as they would normally do in regular daily activities.
Volunteers were allowed to go into the washroom unescorted (to prevent any observational bias) and given up to 2 minutes to complete their handwashing activities (with non-antimicrobial soap). Upon return, volunteers were escorted back to the test area where they were asked to put the three fingertips of the left hand onto a separate petri dish for a period of 30 seconds. After the 30 seconds were complete, the petri dish was labeled with i) the initials of the volunteer; ii) the letters AF for after washing; and iii) the designation AB or C. Again, the volunteers were asked to press firmly to ensure that an impression of the fingerprint would be left behind. Upon completion of the test, the plates were incubated at 98.6ºF (37ºC) for 24 hours.
Bacterial enumeration and calculations
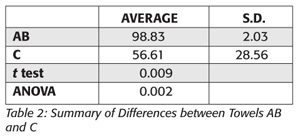
After 24 hours, the plates were removed from the incubator and colonies were enumerated. Numbers were checked by a second person and then entered into Microsoft Excel. Differences between the B4 and AF plates, represented as percent reduction were then calculated. All AB and C percent reductions were then pooled and assessed for significant difference by both the Student’s t test with unequal variance as well as the single factor analysis of variance (ANOVA).
Results
Visual confirmation of fingerprint source of bacteria
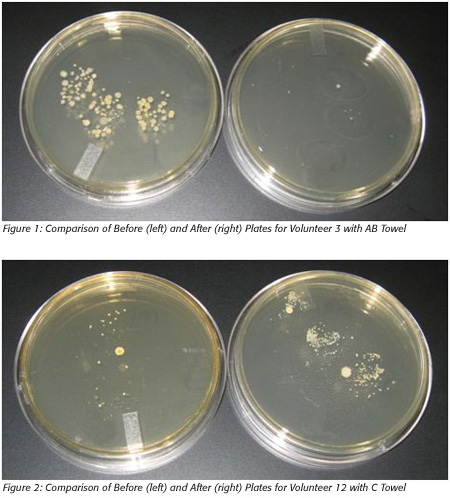 A total of 14 volunteers completed the test as required. Seven were given the AB towel and seven were asked to use the conference-provided towel.After 24 hours of incubation, the plates were visually inspected for evidence of bacterial colonies associated only with the fingerprints left by the volunteers. Figures 1 and 2 provide prototypical examples of the association of fingerprints and bacterial colonies.
A total of 14 volunteers completed the test as required. Seven were given the AB towel and seven were asked to use the conference-provided towel.After 24 hours of incubation, the plates were visually inspected for evidence of bacterial colonies associated only with the fingerprints left by the volunteers. Figures 1 and 2 provide prototypical examples of the association of fingerprints and bacterial colonies.
AB vs. Conference towel
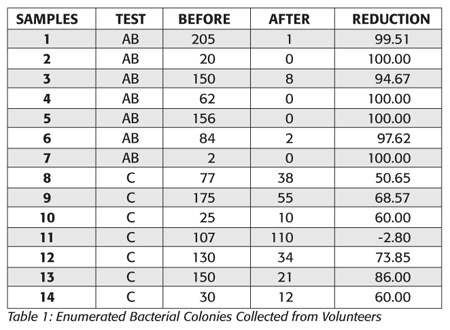 Table 1 provides the raw data collected from the test, whereas Table 2 offers a summary of the results including the p-value of the statistical analysis of the data by both the single-tailed Student’s t test with unequal variance and single factor ANOVA.
Table 1 provides the raw data collected from the test, whereas Table 2 offers a summary of the results including the p-value of the statistical analysis of the data by both the single-tailed Student’s t test with unequal variance and single factor ANOVA.
Discussion
The aim of this study was to determine any improvements in hand hygiene using the AB towel over the C towel provided at the conference. Moreover, by performing the study in the field at an open conference, the potential for any bias was completely removed. Based on the visual evidence, there was an improved reduction between the AB and the C towels. Enumeration and pooling of the results support the visual observations as AB offered a statistically significant improvement in the reduction of bacteria from the fingertips of volunteers. The results demonstrated consistently a 40–50% increase in the efficacy of handwashing compared to the C towel.
In one case, volunteer 11, there was a slight increase in the bacteria on the hands after the handwashing process and use of the C towel. This suggests that the means by which this individual performed hand hygiene was insufficient to remove the bacteria that were on the hands.
The calculated means of percent reduction were 98.83±2.03 and 56.51 ± 28.5 for AB and C regular paper towel respectively, further illustrating the relatively small range of variance for AB in its activity.
There were several observations made concerning the usefulness of AB in encouraging individuals to perform handwashing. Several of the volunteers after washing their hands verbally shared their preference of the ‘green’ towel over the traditional white towels provided. When queried further, answers varied but centered around two specific themes. The first was the color itself, which differed greatly from the standard white and brown colors that are the staple of most paper towels. The second was the apparent feel of the towel as shared by those who used the AB towel. Post drying, people suggested their hands ‘felt’ cleaner although when asked for reasons, none were offered. A further question to these individuals was asked to determine whether they had an increased interest in washing their hands upon observation and knowledge of the AB towel and the response was unanimously positive.
There are considerations that have to be taken into account with this field test. While the number of volunteers tested was large enough to support statistical testing, there is a need for a more comprehensive trial. In addition, to best mimic the field, many parameters involved in clinical human testing, including observation of the handwashing process were not performed. This choice stems from the fact that observation is known to lead to the Hawthorne effect,[8] in which individuals will act in a manner that is different from routine in order to please the observers. This study was aimed at investigating improvements in the field and as such, observation was considered to present bias.
In addition, by not initially contaminating the hands with a set number of bacteria, the ability to identify 4 log10 reduction, which is a mandate of ASTM fingerpad tests,[9] was not possible. Lucet et al.[10] investigated the natural contamination of hands and found that through normal daily activities, there was traditionally less than 3 log10 bacteria. This study correlated with this current study in which the level of contamination of the hands never rose about 1000.
Another consideration is associated with the potential for hand contamination during travel from the handwashing area to the testing area. The AB product has been shown to exhibit persistence in the laboratory environment (Kyle McGovern, Bioscience Laboratories) and may have acted to prevent contamination of the hands post-washing, thus creating bias. Yet, as this was an investigation of the field in which any contamination would be considered to be worthy of note, these results only show the efficacy of the AB product both during and after the handwashing process.
The results of this study suggest testing the field without observational bias may reflect poorly on the handwashing process. This observation is not new; however, as Bottone et al.[11] demonstrated a similar dissimilarity between the laboratory-based activity of lotion soap and its effectiveness in the field. In this case, the soap may not have performed any less effectively, but the lack of proper adherence to hand hygiene reduced the overall effectiveness of the product.
Conclusion
Based on the results of this trial, the use of AB in the handwashing process resulted in significantly higher (p=0.009) bacterial reduction than the use of the towel provided at the conference. Furthermore, this study demonstrates the lack of proper handwashing compliance in the study population. This second observation is not new as handwashing practice has been in question for some time[12] and continues to be a significant issue in the prevention of infection spread. The use of an antibacterial towel has been shown in the laboratory environment to improve the reduction of bacteria during the handwashing process. This study demonstrates that the use of this towel also improves the safety of hands in the field. The use of antibacterial towels should therefore be encouraged in areas where infection control is mandatory including the healthcare, food and travel industries.
Jason A. Tetro, B.Sc., Ph.D. (Hon.) is at MI-SCI Consulting and Communications. For more information, please visit www.cascades.com.
References
1. Erasmus V, Daha TJ, Brug H, Richardus JH, Behrendt MD, Vos MC, van Beeck EF. 2010. Systematic review of studies on compliance with hand hygiene guidelines in hospital care. Infect Control Hosp Epidemiol 31(3): 283–294.
2. Soon JM, Baines R, Seaman P. 2012. Meta-analysis of food safety training on hand hygiene knowledge and attitudes among food handlers. J Food Prot 75(4): 793–804.
3. World Health Organization. 2009. WHO Guidelines on Hand Hygiene in Health Care: A Summary.
4. Nevo I, Fitzpatrick M, Thomas RE, Gluck PA, Lenchus JD, Arheart KL, Birnbach DJ. 2010. The efficacy of visual cues to improve hand hygiene compliance. Simul Healthcare 5(6): 325–331.
5. Gould DJ, Drey NS, Creedon S. 2011. Routine hand hygiene audit by direct observation: Has nemesis arrived? J Hosp Infect 77(4): 290–293.
6. Todd EC, Michaels BS, Holah J, Smith D, Greig JD, Bartleson CA. 2010. Outbreaks where food workers have been implicated in the spread of foodborne disease. Part 10. Alcohol-based antiseptics for hand disinfection and a comparison of their effectiveness with soaps. J Food Prot 73(11): 2128–2140.
7. Sattar, SA. Allen Denver Russell Memorial Lecture, 2006. The use of microbicides in infection control: a critical look at safety, testing and applications. J Appl Microbiol 101(4): 743–753.
8. Kohli E, Ptak J, Smith R, Taylor E, Talbot EA, Kirkland KB. 2009. Variability in the Hawthorne effect with regard to hand hygiene performance in high- and low-performing inpatient care units. Infect Control Hosp Epidemiol 30(3): 222–225.
9. ASTM International. 2010. ASTM E2276 - 10 Standard Test Method for Determining the Bacteria-Eliminating Effectiveness of Hygienic Handwash and Handrub Agents Using the Fingerpads of Adults. West Conshohocken, PA: ASTM.
10. Lucet JC, Rigaud MP, Mentre F, Kassis N, Deblangy C, Andremont A, Bouvet E. 2002. Hand contamination before and after different hand hygiene techniques: A randomized clinical trial. J Hosp Infect 50(4): 276–280.
11. Bottone EJ, Cheng M, Hymes S. 2004. Ineffectiveness of handwashing with lotion soap to remove nosocomial bacterial pathogens persisting on fingertips: A major link in their intrahospital spread. Infect Control Hosp Epidemiol 25(3): 262–264.
12. Gilmour J, Hughes R. 1997-1998. Handwashing: still a neglected practice in the clinical area. Br J Nurs 6(22): 1278-1280, 1282–1284.
Looking for quick answers on food safety topics?
Try Ask FSM, our new smart AI search tool.
Ask FSM →






