In Pursuit of Clean Packaging with No Chemicals of Concern
The food industry is just beginning to address PFAS derivatives as EPA has done, but it is making slow progress

The food packaging industry has its hands full with focus on improved supply chain resiliency, sustainability pulls toward store-drop-off flexible film recycling, and food waste prevention. While these are important initiatives, clean packaging—a nice "to-do" about 15 years ago—is now a "must-do." The good news is that action is possible and is in alignment with efforts to move the dial on climate change and create a more sustainable food system.
While clean packaging is moving to the top of the agenda for many brands and packaging converters, it is important to recognize that since we have "kicked the can down the road" on clean packaging for many years, a high level of commitment and resources is now needed. Now is the time for action. We can regain focus on the first function of packaging: to protect food. This is a fast-moving area, and this summary provides a glimpse into the severity of the issue and offers hope that we can work together to make the world a better place. This summary covers the background, addresses why action is needed now, and uses per- and poly-fluoroalkyl substances (PFAS) and phthalates as examples of the complexities. The final section covers recommendations for brands and packaging converters.
Timeline from 1958 to 2022
We have accepted that packaging interacts with food for at least 63 years—beginning in 1958, when the Food Additive Amendment stated that packaging was considered an indirect additive and continuing with similar actions by other governing bodies. Over the years, this has been very loosely interpreted primarily because of:
- Increased attenuation of equipment resulting in detection of parts-per-trillion versus the parts-per-hundred that was measurable in 1958
- A more refined understanding of migration from packaging with, for example, high-fat foods having higher migration rates from packaging than foods with no fat
- A nuanced approach allowing the use of additives for specific applications based on length of contact time, temperature of exposure, and what constitutes a functional barrier.
So, one additive may be approved for use with water treatment prior to bottling and not approved for use with soup retort processed in a can. To assess how much of an additive is allowed, FDA and other global governing bodies use food-simulating liquids (FSLs) to mimic foods such as soup and water, and to measure migration.
Fast-forward to 2022: Over 10,000 chemicals are now approved for direct food-contact packaging worldwide, and many of these intentionally added substances (IAS) and non-intentionally added substances (NIAS) are a concern.1 Of current concern are:
- Phthalates—predominantly used with PVC, metal closures, and other packaging
- BPA—predominantly used with polycarbonate bottles and metal cans
- PFAS—predominantly used with grease-proof coatings on paperboard
- Perchlorate—predominantly used in business-to-business dry ingredient liners.
Clean packaging now means that that packaging does not contain any chemicals of concern. Chemicals of concern are chemicals that adversely impact human and environmental health. They are divided into Tiers 1, 2, and 3, and can be found in a downloadable spreadsheet.2
Clear Rationale for Coordinated Action Now
Food packaging uses chemicals to convert raw materials (bauxite, iron, fossil fuels, biomass, trees, sand, etc.) via blow molding, blown and cast extruding, laminating, and printing into consumer-ready food packaging materials. The need for coordinated action on chemicals of concern used in converting is due to:
- Increased Legislation: Recent bills are underway to prompt the reevaluation of approved chemicals of concern and to create a complex web that is tough for industry to navigate. For example, in the U.S.:
- Schakowsky (Illinois) introduced the Food Chemical Reassessment Act of 2021 (FCRA), which would, under the direction of the Office of Food Safety Reassessment, reassess 10 chemicals of concern including PFAS, phthalates, and BPA
- The Toxic Free Food Act, introduced by DeLauro (Connecticut) would require FDA to close the so-called GRAS loophole, which allows manufacturers to designate such chemicals as "generally recognized as safe" (GRAS), without FDA review or public notice, and make the industry's chemical food additives subject to FDA approval
- The Coalition of Northeastern Governors (CONEG) has been a testing ground for other states on packaging legislation since 1992; in 2021, the Model Toxics in Packaging Legislation was revised to include PFAS, phthalates, and other chemicals in packaging
- More than 200 state policies have been enacted to address chemicals of concern; Safer States carries the database of all bills.3
- Improved science: Data-based and agreed understanding of the cancerous and endocrine disruption and bioaccumulation associated with many approved chemicals highlights the need to reconsider authorizations. Scientific material science has defined or can define safer alternatives, along with the agreed understanding that chemicals of concernare an issue.4
- Obvious data gaps: Packaging disposal methods such as composting allow components within the packaging to be released into the environment. For example, few of the many chemicals that exist in compostable packaging are measured by the compostability standards in place, and PFAS has only been measured as a part of this standard for the past two years.
- Serious end-of-life concerns: Some approved chemicals cannot be removed from packaging to enable safe and economical reuse, recycle, and degradation of packaging. When a container is reused for a different product, we may have an additive in a formerly approved high concentration now in contact with a food with which it was not designed to be in contact. Recycled packaging varies in its ability to screen out additives and ensure that only what is allowed for direct food contact is, in fact, in direct food contact.5 Furthermore, if biodegradation occurs, a situation can arise in which chemicals used are released as the packaging degrades. This results in an expanded array of contaminated resources used in producing our food supply. Land and water are contaminated by these "forever chemicals" that build up in our bodies and never break down in the environment.
Chemicals of Concern Complexities Illuminated by PFAS
In 1965, PFAS chemicals began to be approved. Notably, per fluorinated octanoic acid (PFOA) was approved by FDA for food contact because it was assumed that no migration would occur into food. The next year, a PFAS proposal was first rejected and then approved by FDA, although data showed that PFAS did migrate from packaging into food, and a longer-term health study on the impact was not conducted nor required.6,7 Oddly, in the next two years, approvals for PFAS were granted.
The Long and the Short
In 2011, long-chain PFAS began to be voluntarily phased out. In 2016, FDA banned the use of long-chain PFAS. Short-chain PFASs were approved without considering biopersistence within the body. Short-chain versus long-chain PFAS were then used for grease-proof paper-based packaging. However, nine years after their authorization, safety concerns over the bio-persistence and higher toxicity of short-chain PFAS 6:2 fluorotelomer alcohol (6:2 FTOH) arose.
Looking for quick answers on food safety topics?
Try Ask FSM, our new smart AI search tool.
Ask FSM →
The Damage
PFAS and its derivatives, PFOA and perfluorooctanesulfonic acid (PFOS), accumulate in humans. Epidemiology studies indicate that exposure can cause cancer and reproductive, developmental, cardiovascular, liver, kidney, and immunological issues. The focus has been on the environment, as environmental costs are high, as well.
In 2021, 3M agreed to pay $850 million in a lawsuit associated with PFAS contamination. This would make contaminators accountable for cleanup. As a solution, the Environmental Protection Agency (EPA) has proposed enveloping PFAS within the Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA), also known as "Superfund Hazardous Substances."
Action on PFAS
The food industry is just beginning to address PFAS derivatives as EPA has done, but it is making slow progress. Maricel Maffini, an independent consultant who has advocated for responsible use of direct food chemicals for 12 years, elaborates, "In 2018, FDA research showed that short-chain PFAS were bioaccumulative. In 2021, the FDA agreed to an industry phase-out of short-chain PFAS compounds that contain 6:2 fluorotelomer alcohol (6:2 FTOH). Thus, by 2024, food packaging suppliers will voluntarily phase out all PFAS, and the FDA is slated to develop a verification process. To wrap things up with PFAS, the FDA needs to reassess the safety of all authorized PFAS still allowed in food contact materials."
Furthermore, PFAS should be considered a chemical class.8 Removal of PFAS from packaging will be accomplished earlier due to the first of at least 15 statewide PFAS bans in food packaging going into effect in December 2022. Anecdotally, an addition to the National Defense Authorization Act banned the use of PFAS in military Meals Ready-to-Eat (MREs) packaging after October 1, 2021.9 FDA, however, has not banned the use of PFAS in direct contact with food packaging. Over 1,235 PFAS lawsuits were filed in 2021. On farms, some PFAS contamination comes from packaging containing PFAS that is composted on farm sites, as well as biosolids derived from sewage sludge from wastewater treatment and industrial waste.10
Phthalates Showcase the Disconnected Science with Many Chemicals of Concern
Food packaging uses phthalates as plasticizers, binders, coating agents, defoamers, and in gasket closures. Although the Consumer Product Safety Commission (CPSC) banned some phthalates for use in children's toys and other articles in 2014 and 2018 because they are anti-androgenic and can interfere with the production and function of male hormones, FDA approval for the following nine phthalates still exists:
- Diallyl phthalate, CAS No. 131-17-9 (DAP)
- Diethyl phthalate, CAS No. 84-66-2 (DEP)
- Butyl phthalyl butyl glycolate, CAS No. 85-70-1 (BPBG)
- Diisooctyl phthalate, CAS No. 27554-26-3 (DIOP)
- Ethyl phthalyl ethyl glycolate, CAS No. 84-72-0 (EPEG)
- Di(2-ethylhexyl) phthalate, CAS No. 117-81-7 (DEHP)
- Dicyclohexyl phthalate, CAS No. 84-61-7 (DCHP)
- Diisononyl phthalate, CAS No. 28553-12-0 (DINP)
- Diisodecyl phthalate, CAS No. 26761-40-0 (DIDP).
Maffini explains, "This is complex and nonsensical. Notably, the FDA agreed to remove the approval for use of 19 phthalates because industry self-reported that they are not in use anymore. FDA didn't opine on whether the uses are safe or not. So, a company can claim a use is GRAS since there is no definitive ruling that phthalates are unsafe. Also, a self-declared GRAS phthalates use does not have to be reported and can be used in food contact packaging."11 This is of particular concern with the supply chain disruption the packaging industry faces and the lack of chain of custody between each converter and the brands. Sadly, this has occurred as recently as 2021, when phthalates thought not to be in use were found on food packaging.12
FDA established a voluntary GRAS notification process in 1997 that essentially allows companies to determine that "the substance is generally recognized, among qualified experts, as having been adequately shown to be safe under the conditions of its intended use," and therefore GRAS. Since then, at least 1,000 new substances have avoided the prior food additive petition process to review their safety.13
Recommendation 1: Take High-Impact Action Fast
Measuring chemicals of concern in food in direct contact with packaging should be in all quality assurance and packaging research and development budgets. While not testing allows both packaging converters and brands to declare, "The presence of a specific chemical was unknown," this is irresponsible and unwise risk management since ignorance that a chemical that is known to be unsafe or illegal at levels used in packaging does not provide sufficient legal or consumer protection. We have seen this to be true recently with BPA, phthalates, and PFAS. Both packaging and food require testing.
To maximize impact, time, and money, use an initial three-step process:
- Identify packaging with a high likelihood of containing these current chemicals of concern. Packaging of concern is packaging with a high likelihood of containing phthalates, BPA, PFAS, and perchlorate, which are the subject of restrictions and consumer concern. Importantly, this includes processing aids, inks, and adhesives.
- Test packaging and food that are highly likely to contain these current chemicals of concern. Importantly, incoming packaging, as well as the finished packaged food, should be tested because packaging processes involving abrasion associated with filling operations—such as vertical-form-fill-seal—can result in an extended shelf-effect migration from packaging into food on a macro- and micro-level. Moving forward, correlations between the amount of chemicals in packaging and the final food consumers consume can be made. In addition, the use of food simulating liquids (FSLs) allows for more rapid initial work that highlights areas of concern.
- Act and plan forward. Replace these current chemicals of concern and set up a protocol testing plan within standard operating procedures to ensure that they do not reenter the packaging system through suppliers, manufacturing locations, or employee turnover.
Phthalates, BPA, PFAS, and perchlorate are the "tip of the iceberg." A prioritized list of 608 chemicals from a compilation of 67 global regulations is available; within these, Tier 1, 2, and 3 chemicals provide more focus. Importantly, this information is available in a database ready for use. In 2021, Packaging Technology & Research was instrumental in defining the 608 chemicals in use in specific food packaging. Some industries have defined a narrower scope. For example, in foodservice ware, the GreenScreen Standard developed by the Center for Environmental Health (CEH) and Clean Production Action (CPA) is a good place to start.14 The above three process steps are then applied to continue the process toward clean packaging.
Recommendation 2: Use Clean Packaging to Build Shared Value
Since 2017, consumers have ranked chemicals in food as their top concern (of greater concern than pathogens), according to surveys conducted by IFIC Food and Health.15 Package suppliers and brands that ensure the absence of unapproved chemicals and Tier 1, 2, and 3 chemicals of concernin their packaging and their food can find shared value. From a shareholder point of view, this is also smart business, as evidenced by the decline of consumers purchasing food contained within packaging that does not declare "BPA free."
The fallout from when consumers and researchers send products and packages for testing, and chemicals that should not be found in the package and food are detected, is tough to combat. Consumer Reports tested 100 food service packaging components and found PFAS. McDonald's, in particular, is accused of violating its food safety pledge. The industry understands that chemicals of concern should not be in direct contact with food. For example, in 2018, the Food Safety Alliance for Packaging within the Institute of Packaging Professionals outlined a list of recommendations.4
When the value for the removal of chemicals of concern is communicated up and down the value chain, trust increases, and the need for policing and testing declines. This saves industry dollars in testing and leaves more space for innovation in replacements, as well as overall package innovation. Packaging suppliers will focus business on brands that value clean packaging. Brands will award business to suppliers that invest in Tier 1, 2, and 3 chemical removal plans. From a retailer view, ensuring that private-label brands do not contain Tier 1, 2, and 3 chemicals of concern creates alignment with like-minded co-manufacturers and packaging suppliers.
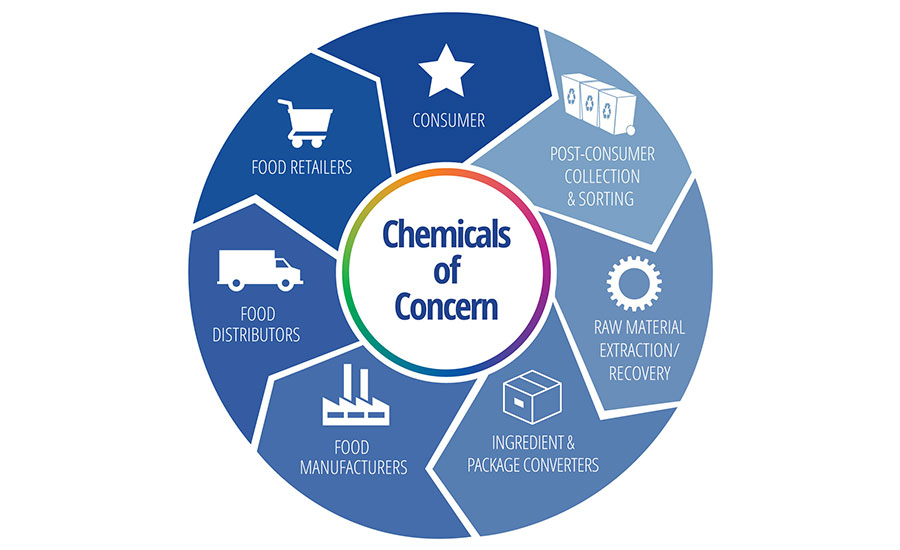
Clean packaging is an opportunity to regain trust in packaging. It is irresponsible and unethical to move toward a two-tiered packaged food supply, in which foods commonly purchased by consumers in a higher economic bracket do not contain chemicals of concern and the opposite is true for the lower bracket. A lower-economic-bracket consumer does not exhibit lower effects from chemicals of concern. To build trust, brands and packaging converters must protect all consumers by ensuring that the use of recyclable, compostable, and reusable packaging is free from contamination across borders. Eliminating chemicals of concern in all global operations and sales of packaging and packaged goods allows for less impeded commerce (Figure 1).
FIGURE 1. Eliminating Chemicals of Concern in All Global Operations and Sales of Packaging and Packaged Goods Allows for Less Impeded Commerce (image by Michela Emmerich; used with permission by Packaging Technology & Research)
Recommendation 3: Fund Pragmatic Research
To ease the worldwide burden of much-needed research, science can be extended across states, borders, and continents. New FDA funding to address food chemicals; $7 million for emerging chemical and toxicology issues, and $11 million for maternal and infant health and nutrition will assist. In addition, funding in other regions is increasing as the issue of chemicals of concern gains momentum.
The work that needs to be done can be optimized much in the same way that EU scientists developed uniform research methods in the early stages of the EU. This is an opportunity for food packaging research conducted at institutes and universities to advance and become more coordinated and industry-oriented. For example, to protect all consumers, when the use of chemicals is eradicated based on rigorous science in a certain region, these chemicals should also be removed from the packaging in other countries. To further maximize research applications, the test design can be reviewed and refined by governing bodies, prior to the research being conducted, to ensure that results can be pragmatically applied.In this way, research conducted in other regions can be funded and then applied worldwide to lower the costs of assessing the suitability of chemicals in direct contact with food. Notably, the exposure for population segments based on age, gender, and chronic disease can be better understood.
Applying systems thinking to where chemicals enter the food system and their toxicological impact will help a number of factors be better understood:
- Interactive reactions: Reactions between chemicals within packaging can occur during the manufacturing of multilayer packaging, forming processes, and for food processing (such as retort cans) in the package. "It is essential to define these reactions and then test for the biological significance of the reaction products so we can get closer to the reality of what consumers are exposed to in direct contact with food packaging," states Maffini.
- Toxicological impact: While the amount of chemicals of concern is certainly relevant, the toxicology measuring the impact is most relevant. Toxicology systems thinking encompassing human physiology and, specifically, infant physiology is needed to connect chemicals of concern to the impact on endocrine disruption, cancer, and other avoidable illnesses. To more accurately estimate human exposure to a food ingredient, FDA and companies use food consumption data from the "What We Eat in America" survey, which is the USDA-based dietary component of the National Health and Nutrition Examination Survey (NHANES). However, this survey does not contain an exposure estimate specific to packaging.16 Furthermore, recommendations from the Select Committee on GRAS Substances (SCOGS) can be followed to address this issue.17,18
- Cumulative effect: It is established and required that entire classifications of chemicals be considered in terms of their cumulative effects, which is required by FDA in 21 CFR 170.18.19 Recently, the European Food Safety Authority (EFSA) was directed to formulate a data-based opinion on phthalates with known endocrine disruptions and then to establish a tolerable daily intake for four of the five phthalates. This means that the combined amounts of the phthalates would be considered.
In Pursuit of Clean Packaging with No Chemicals of Concern
Like many readers, I am proud to work in the food packaging industry. I will be prouder if we invest in the concerted action needed to make the world a better place by addressing chemicals of concern in the pursuit of clean packaging. We can, and we should.
References
- Groh, K. J., B. Geueke, O. Martin, M. V. Maffini, and J. Muncke. "Overview of intentionally used food contact chemicals and their hazards." Environment International 150 (May 2021). https://doi.org/10.1016/j.envint.2020.106225.
- "Tiers 1, 2, and 3 Chemicals of Concern." 2022. https://docs.google.com/spreadsheets/d/1Eu9qbzk5gPST39ppdmM8PwsYqdm4WEVjL2oWHiOijw0/edit#gid=593236777.
- Safer States. "States Lead the Way." 2022. https://www.saferstates.com/states-in-the-lead.
- Institute of Packaging Professionals (IoPP) and Food Safety Alliance for Packaging (FSAP). "Food Packaging Product Stewardship Considerations." March 9, 2018. https://www.iopp.org/files/Food Packaging Product Stewardship Considerations FSAP-IoPP v1_0.pdf.
- U.S. Food and Drug Administration (FDA). "Guidance for Industry: Use of Recycled Plastics in Food Packaging (Chemistry Considerations)." 2021. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-use-recycled-plastics-food-packaging-chemistry-considerations.
- FDA. "FDA memorandum on Zonyl 'pin down extent of use.'" February 3, 1966. https://www.documentcloud.org/documents/21297031-fda-memo-on-zonyl-pin-down-extent-of-use.
- FDA. "FDA memorandum determining use of Zonyl is safe." July 21, 1966. https://www.documentcloud.org/documents/21295354-fda-memo-determining-use-of-zonyl-is-safe.
- Kwiatkowski, C., D. Q. Andrews, L. S. Birnbaum, et al. “Scientific Basis for Managing PFAS as a Chemical Class.“ Environmental Science and Technology Letters 7, 8 (2020): 532–543. https://doi.org/10.1021/acs.estlett.0c00255.
- 116th Congress. "National Defense Authorization Act for Fiscal Year 2020." December 20, 2019. https://www.congress.gov/bill/116th-congress/senate-bill/1790/text.
- Clayton, Chris. "'Forever Chemicals' and Risks to Farms." Progressive Farmer. May 9, 2022. https://www.dtnpf.com/agriculture/web/ag/livestock/article/2022/05/06/michigan-farm-cautionary-tale-pfas.
- FDA. "Indirect Food Additives: Adhesives and Components of Coatings; Paper and Paperboard Components; Polymers; Adjuvants, Production Aids, and Sanitizers." Federal Register. May 20, 2022. https://www.federalregister.gov/documents/2022/05/20/2022-10531/indirect-food-additives-adhesives-and-components-of-coatings-paper-and-paperboard-components.
- Loria, K. "Dangerous PFAS Chemicals Are in Your Food Packaging." Consumer Reports. March 2022.
- Neltner, T., N. R. Kulkarni, H. M. Alger, M. V. Maffini, E. D. Bongard, N. D. Fortin, and E. D. Olson. "Navigating the U.S. Food Additive Regulatory Program." Comprehensive Reviews in Food Science and Food Safety 10 (2011): 342. https://doi.org/10.1111/j.1541-4337.2011.00166.x.
- Green Screen. "GreenScreen Certified™ Standard for Food Service Ware." 2022. https://www.greenscreenchemicals.org/certified/food-service-ware.
- Food Insight. "2021 Food & Health Survey." May 19, 2021. https://foodinsight.org/2021-food-health-survey/.
- Maffini, M. V., H. M. Alger, E. D. Olson, and T. G. Neltner. "Looking back to look forward: A review of FDA's food additives safety assessment and recommendations for modernizing its program." Comprehensive Reviews in Food Science and Food Safety 12 (2013): 439–453.
- FDA. "History of the GRAS List and SCOGS Reviews." January 4, 2018. https://www.fda.gov/food/gras-substances-scogs-database/history-gras-list-and-scogs-reviews.
- FDA. "GRAS Substances (SCOGS) Database." March 3, 2022. https://www.fda.gov/food/generally-recognized-safe-gras/gras-substances-scogs-database.
- Code of Federal Regulations. 21 CFR 170.18: "Tolerances for related food additives." https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=170.18.
Claire Koelsch Sand, Ph.D. is CEO of Packaging Technology & Research LLC, as well as an Adjunct Professor at both California Polytechnic State University and Michigan State University. She has over 35 years of experience in food packaging. She ranks innovative packaging science solutions to extend shelf life and align with consumer needs, generates implementation roadmaps, and aligns business cases. Dr. Sand is an IFT Fellow and a Riester-Davis-Brody Lifetime Achievement in Food Packaging Award recipient. She holds a Ph.D. in Food Science and Nutrition from the University of Minnesota, as well as M.S. and B.S. degrees in Packaging from Michigan State University.








