Dealing With Dioxin: The State of Analytical Methods
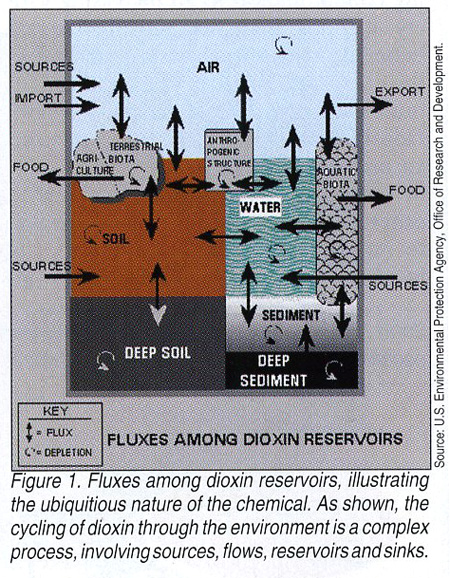
On June 12, 2000, the U.S. Environmental Protection Agency (EPA) reported that its nine-year assessment of dioxin establishes the chemical as a carcinogen and that the risk posed by exposure to dioxin and dioxin-like compounds is greater than previously believed. The agency stated that the vast majority of dioxin exposure to Americans is found in the foods we consume, and controversially, that there is a direct link between fatty foods and dioxin residues. Specifically, EPA estimates that most dioxin exposure occurs through the diet, with more than 95°/o of dioxin intake for a typical person coming through dietary intake of animal fats. Small amounts of exposure occur from breathing air containing trace amounts of dioxin on particles and in vapor form, from inadvertent ingestion of soil containing dioxin, and from absorption through the skin contacting air, soil or water containing minute levels (Figure 1. Fluxes among dioxin reservoirs, illustrating the ubiquitous nature of the chemical. As shown, the cycling of dioxin through the environment is a complex process, involving sources, flows, reservoirs and sinks.).
Dioxins are not created intentionally, but they can be produced through industrial processes, including combustion, chlorine bleaching of pulp and paper and through certain types of chemical manufacturing and processing. These processes result in widespread, low-level exposure of the general population to dioxins. Since the chemical is associated with several lymphomas and lung cancer, new concerns about the safety of the domestic food supply have set off renewed debate about how to assess the potential hazards and risks of dioxins.
As a result of improved scientific understanding about the mechanisms of dioxin toxicity, significant new studies of dioxin’s carcinogenic potential in humans and increased evidence of other adverse health effects, EPA announced a schedule for scientific and public comment as a final stage in the process of reassessment of dioxin exposure and associated human health risks. The final dioxin reassessment will consist of three parts, with the latter two draft documents, “Part II: Chapter 9: Toxicity Equivalence Factors for Dioxin and Related Compounds,” and “Part III: Integrated Summary and Risk Characterization for 2,3,7,8-Tetrachlorodibenzo-p- Dioxin and Related Compounds” undergoing external peer review this summer. A rolling public comment period commenced following the September 2000 Scientific Advisory Board review of revised drafts.
Recently the National Academy of Science (NAS) conducted a study that focused on microbial, chemical and physical hazards of foods. In August of 1998, NAS released a report that made a number of recommendations that centered on development of a national food safety program. This report led to an executive order by President Clinton creating the Council on Food Safety. One of the areas being addressed by the Council is chemical contamination of food. Recent incidents of chemical contamination of the food chain have raised public awareness of the potential for contamination of the human diet to have long-term effects. Dioxin and dioxin-like compounds are among the more high-profile contaminants found in the environment and in food. These compounds have been involved in a number of incidents, such as the “Yusho” (oil disease) in Japan, the “Yu-Cheng” incident in China, and the catfish incident in the U.S.
The most recent incident involved accidental contamination of feed with transformer oil containing polychlorinated biphenyls (PCBs) and dioxins in Belgium. The result was the contamination of meat and food with PCBs and dioxins, which occurred in recycled animal fat used to produce feed. Contamination was subsequently found in poultry and poultry products, pigs and cattle. All contaminated animals and animal products were destroyed. After the Belgium incident, tolerance levels for PCBs and dioxin contamination in milk was set at 100 pg/kg of fat (100 ppb) for PCB and 5 picograms TEQ per gram of fat (ppt) for dioxins. The European Union is currently evaluating the Belgium data as part of its efforts to set tolerance limits. The direct costs associated with this incident have been estimated at $0.6 to $1.1 billion.
Chemistry
Polychlorinated dibenzodioxins (PCDDs), polychiorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) constitute a group of polyhalogenated aromatic compounds that have become known as “dioxin” and “dioxin- like compounds.” Dioxins and PCBs are considered to have a number of toxic effects that include dermal toxicity, immunotoxicity, reproductive impacts, endocrine disruption and carcinogenicity. Dioxins and PCBs have many chemical properties in common. The most notable characteristic of these compounds is the fact that they are very stable and resist breakdown from heat, acids and bases. This stability allows them to bioaccumulate. Dioxins and PCBs are believed to cause human health effects through similar mechanisms.
There are more than 445 specific isomers in these three groups of compounds. Of the 445 isomers, 30 compounds have been identified as having a significant health risk to humans. The actual assumed toxicity of the individual isomers varies over an extremely wide range. Of the isomers defined as being significantly toxic, 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) is considered the most toxic of all isomers. The EPA, World Health Organization (WHO) and other regulatory entities have developed a set of weighting factors called toxic equivalent factors (TEFs) that relate the level of toxicity of each of the isomers to the toxicity of 2,3,7,8-TCDD.
These compounds are very lipophilic and are known to bioaccumulate in the food chain. The primary pathway for exposure in humans is considered to be the diet. However, the primary pathway into the human diet is believed to be through atmospheric deposition. Or, put another way, dioxin and PCB contamination of food is a by-product of the environmental distribution of these compounds. Residues of dioxins and PCBs have been found at various levels in fish, sediments, sludge, soils and air samples analyzed as part of individual and collaborative studies conducted in a variety of countries. Dioxins and PCBs have also been found in the milk, adipose tissue and blood serum samples from humans from all parts of the globe.
Methods for Trace Analysis
Dioxin and PCBs have been found at toxic at levels in the part per trillion (ppt) range. To put this into perspective, the 1 ppt level is analogous to looking for a BB in two train carloads of wheat. The level of detection needed for dioxins make the current methods for the analysis of these compounds tedious, difficult to perform and expensive. The availability of labs to perform the analysis is limited due to the capital investment and knowledge necessary to meet stringent detection limit and the quality control parameters that are typically mandated.
The current analytical approach utilizes an analytical technique known as isotopic dilution. Isotopic dilution refers to a technique where a known amount of the carbon-13 form of a compound is spiked into the sample. This establishes a solution with chemically identical forms of the compound that can be separately quantified on a mass spectrometer. This type of method is isomer specific, very sensitive and robust. Isotopic dilution is one of the most quantitative analytical methods in use today. This type of analysis has two main parts, the extraction/separation (isolation of the compounds of interest from the matrix) of the sample, and the instrumental analysis.
The analytical part of the dioxin and congener-specific PCB methods utilizes gas chromatography (GC) with high resolution mass spectrometry (HRMS) detection. The advantage of the mass spectrometer is its ability to distinguish between the isotopic forms of the elements due to their differences in atomic mass. An additional advantage is their sensitivity. This allows the ratio between each analyte and its associated isotopically labeled standard to be determined without the inaccuracies that are associated with measuring the two concentrations in separate runs. The accuracy of the overall analysis depends only on the ability to accurately spike the sample, weigh the sample and measure the ratio. This approach allows the lab to quantitate the effects of interferences, correct for minor losses due to sample handling and determine isomer specific concentrations.
The major challenges with regard to the analysis of food products lie in the extraction and separation of the dioxins and PCBs from the other compounds found in the matrix. There are significant differences in the approach to extraction and cleanup needed for fruits, hard vegetables, soft vegetables, grains, dairy products, fish and meats. Foods that combine ingredients into a final product can further complicate the matrix with the chemical additives. Foods and food ingredients such as milks, plant extracts, beef and fish have been routinely analyzed for the presence of dioxins and PCBs, utilizing these techniques at the ppt level.
The extraction phase establishes the relationship between the isotopically labeled standards that are added to the sample and the analytes present in the sample. Once this ratio is set, no chemical or physical process will change the analyte to standard ratio. After spiking the sample with the isotopically labeled standards, a sequence of steps is applied to the sample to separate the dioxins and PCBs from the rest of the matrix. Extraction begins with preparation of the sample, which can be as simple as weighing a specific sample size, or as advanced as using homogenization and disruption of the cell walls of the sample using ultrasonic blenders. Once the sample is prepared for extraction, a known amount of isotopically labeled standard is added. The actual extraction can involve processes as simple as dissolving a known amount of sample in a solvent, to the more complex Soxhlet-type extractions.
After extraction, the raw sample extract will be processed through a series of cleanup steps to remove interfering compounds that may have extracted with the compounds of interest. This phase of the process typically utilizes a variety of chromatographic separation techniques. The more complex the extraction and cleanup for a particular type of sample, the greater the potential for loss of materials of interest. During this part of the process, the recoveries of the analyte and the isotopically labeled standard will remain the same, preserving the ratio and subsequent quantitation of the analytes in the samples.
Sample Screening Methods
Dioxin and PCB contamination is not an issue for all foods. The challenge is to identify the specific food products that are contaminated or are most susceptible to such contamination. Risk-based sampling plans that utilize screening procedures followed by more detailed analysis for positive results offer the most efficient approach for identifying sources of contamination and for managing the risk. There have been significant efforts made toward developing screening assays for determining dioxin and PGB contamination, With a screening assay, speed and cost savings are provided at the expense of specificity. Screening assays must be at least as sensitive as the conventional assay while minimizing the probability of false negatives. Assays of this type provide the upper limit of detection for the target analyte. To determine the actual concentration, a conventional full screen-type analysis would still need to be performed. However, for assessing whether a contamination issue exists, or for identifying critical control points in a process, a screening assay combined with a conventional full screen-type analysis would provide significant cost savings while minimizing the processor’s risk.
Two cost-effective approaches have been developed for screening samples: biotech approaches and instrumental methods. The two approaches utilize different principles for detection. The biotech approach relies on the chemical reactivity of compounds, while the instrumental methods respond to physical properties of the compounds. Both approaches rely on the same basic extraction method, while reducing the cost of the analytical measurement. The biotech approach utilizes immunoassay-type tests and AAH receptor-type tests. In comparison studies, however, the biotech approach has been found to routinely provide an overestimation of the dioxin content of samples. Interfering compounds are one source of false-positives in biological assays. The extraction and separation phase of the analysis is designed to remove interferences; however, there are compounds that may not be removed by the cleanup procedure. In the instrumental approach, interferences can be identified in the raw data, additional cleanup of the extract can be performed and the sample reanalyzed. With the biotech approach, there is no means to determine the effects of interfering compounds.
False-negatives are an even bigger problem for the biotech approach, the risks of which include an increased potential for contaminated products to reach the consumer. This difficulty arises as a result of the inability of the biotech approach to measure recovery from the same sample as the analyte is measured, since internal standards cannot be added to the sample. Labeled standards behave chemically like the analytes, so a biotech approach does not distinguish between the two. If the sample matrix results in analyte loss during extraction and cleanup, the contaminant will not be detected. Performing a recovery measurement from duplicate aliquots of the same sample can minimize the problem of false-negatives due to analyte loss. However, this approach will double the analytical costs.
Conclusion
Research is needed to determine whether there is a cause-and- effect relationship between exposure to dioxin and PCBs and the range of potential adverse human health effects. Evidence has been collected that suggests that the background levels of dioxin and PCBs in the environment do have such an adverse effect. Thus, it is reasonable to expect continued efforts to regulate the levels of these compounds in food and the environment. Trace levels of these compounds have been found in our lakes, streams, rivers, soil, air, plants and animals. Since they are stable compounds in the environment, we expect to see ever-increasing levels. As long as these conditions persist, the probability for contamination of the human diet is high. It is important to minimize the risks to humans and liabilities of the companies preparing and manufacturing food products by developing and utilizing effective risk-based monitoring programs and improved analytical and testing methods.
J. Ronald Haft, Ph.D., is president and CEO, and Franklin M. Stevens is co-director of Analytical Services, for Triangle Labs, Research Triangle Park, NC.
Looking for quick answers on food safety topics?
Try Ask FSM, our new smart AI search tool.
Ask FSM →