Safe to the Last Drop: A First-Hand NCIMS HACCP Pilot Experience
In 1997, the National Conference on Interstate Milk Shipments (NCIMS) appointed a committee of regulatory officials and industry and academic professionals to address how a Hazard Analysis & Critical Control Points (HACCP) system could be implemented, evaluated, monitored and enforced under the NCIMS as a voluntary alternative to the traditional Inspection/Rating/Check rating system used in the dairy industry. (See “Got HACCP?” for information on HACCP as it relates to the dairy industry). The HACCP concept, noted the committee, would enable those operating under and regulating under a HACCP system to move to a preventive approach, whereby food safety hazards are identified and controlled in the manufacturing environment.
As a result, the NCIMS HACCP Committee developed a pilot program for dairy processing plants, which was authorized as a voluntary program at the 1999 NCIMS Conference. The goals of this program, as set forth by the committee, are to:
• Use current National Advisory Committee on Microbiological Criteria for Foods (NACMCF) HACCP principles that are consistent with current U.S. Food and Drug Administration (FDA) HACCP recommendations;
• Continue to assure at least the same level of milk safety provided by the traditional Inspection/Rating/Check Rating System; and to
• Continue to provide uniformity and reciprocity under the HACCP alternative to the traditional Inspection/Rating/Check Rating System used by the dairy industry.
In July 1999, the NCIMS HACCP Committee sent applications for participation in the pilot study to all 50 states. Out of 16 applicants, six were chosen that represent a range of plant sizes, products and FDA regions. The dairy operations selected were: Publix, Lakeland, FL; Tropical Cheese, Perth Amboy, NJ; Michigan Milk Producers Association, Ovid, MI; Garelick Farms of Maine, Bangor, ME; Safeway, Bellevue, WA; and Gossner Foods, Logan, UT. Following baseline sanitation ratings of each facility, advisory visits with FDA and state regulators were performed, training workshops were held, and the selected plants were audited and listed by the state agencies in early 2000.
As noted, Garelick Farms of Maine, a Division of Suiza Foods, was selected as one of the initial six plants to participate in the NCIMS HACCP Pilot Program. Established in 1902 as Grant’s Dairy, Garelick Farms of Maine produces fluid milk and juice products, which it has bottled at its current location in Bangor since 1995. The company markets its products in Maine, New Hampshire and Massachusetts. The company volunteered for the program for several reasons, including a strong background in HACCP planning and implementation, the promise of meeting an interesting challenge, and the desire to be an industry leader. The company also recognized the potential for mandatory HACCP in the future and chose to be proactive in the development of HACCP as an alternative regulatory program for dairy processors. Garelick Farms of Maine has gained many valuable insights as a pilot plant in the NCIMS HACCP program, which will be reviewed in this article.
Setting the Groundwork
Garelick Farms of Maine and the other five pilot sites took several initial steps in implementing the NCIMS HACCP pilot program into their dairy operations, which included the training of teams (both plant and regulatory personnel); advisory visits, team meetings of the plant HACCP team for each operation; documentation and verification of prerequisite programs (PPs); and employee training.
Training of Teams. All six pilot sites and some alternates, along with regulatory personnel, participated in a training session conducted by NCIMS and FDA. The Garelick Farms of Maine team included Gus Peters, Regional Vice President Quality Assurance, Garelick Farms; Steve Pierson, FDA; Audrey Slattery and Glen Mehuren, State of Maine Department of Agriculture; Al Lorenz, Plant Engineer and Rebecca Piston, Quality Control Manager, Garelick Farms of Maine. The purpose of the training session was to provide the same information to all of the participants in the pilot program at the same time. A pilot program document listing the requirements that all participating sites were to follow was provided, and the training included hands-on exercises based on these requirements, such as conducting a hazard analysis and developing prerequisite programs.
Advisory Visits. The same group also met during advisory visits, in which details of HACCP plan development and implementation approaches were worked out by the individual plants with input from the regulatory personnel. The aim of both the training session and the advisory visits was to make this process a cooperative effort between industry and regulators to encourage successful planning and implementation.
Plant HACCP Team Meetings. One of the steps in the dairy HACCP pilot program is to establish a HACCP team. The Garelick Farms of Maine Plant HACCP Team consists of the quality control manager, the plant engineer, the production manager and two production lead- men. It is important that the HACCP team includes the people who are out working on the plant floor on a daily basis because these individuals are most likely to know critical information as to what is actually happening in the plant. This knowledge is critical when the company is going through the process flow and hazard analysis to ensure that what they are trying to manage in the HACCP plan accurately reflects what is actually happening in the plant. Meetings of this on-site team involved the review of process flows and hazard analysis, discussion about the documentation and verification of prerequisite programs, and implementation details customized to the Garelick Farms of Maine plant.
Training of Employees. HACCP basics were communicated plant-wide through group meetings, with one-on-one training for dairy personnel involved with prerequisite programs and specific HACCP related activities.
The actual development of the site-specific NCIMS HACCP plan occurred between November 1999 and January 2000. In the development of the HACCP plan for the Garelick Farms of Maine plant, the company followed the steps outlined in many standard HACCP references. In addition to assembling the plant HACCP team, the preliminary steps of plan development were:
• Describe the food and distribution. In order to conduct a successful hazard analysis and to effectively identify critical control points (CCPs), it is important to know the properties of the food you are trying to manage, because the type of product and the type of processing used to manufacture that food is going to have some bearing on the hazards that will need to be considered.
• Describe the intended use and the consumers of the food. With fresh dairy products, the intended consumers run the gamut from infants to the elderly. The processor needs to be conscious of potential hazards that can affect all of these age groups.
• Develop a process flow diagram. When developing the process flow diagram, keep it as simple as possible. Diagramming every minute detail of the flow makes conducting an effective hazard analysis extremely cumbersome, and detracts from the identification of the critical processing steps. The process flow diagram should include all of the major inputs to the processing operation, such as ingredients, air, water and rework. Figure 1 is an example of a generic process flow diagram for illustrative purposes.
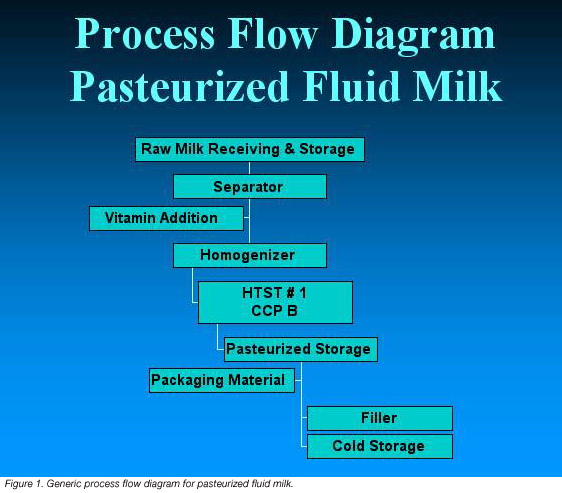
• Verify the flow diagram. This step allows the dairy to confirm that the flow diagram has identified all steps that might impact the hazard analysis.
Identifying Potential Hazards
The hazard analysis stage of developing a HACCP plan is critical to the successful functioning of the plan. During this phase of the Garelick Farms of Maine pilot experience, all phases of the process were considered, from ingredients (including water, air, rework), to processing steps, packaging and storage, through to the intended use of the final product. At each stage, the three types of potential hazards—biological, chemical and physical—were considered and identified as being controlled by a prerequisite program or a CCP. The first determination is based on whether control of the hazard exists in the form of an already established voluntary or mandatory prerequisite program, such a drug residue testing, vitamin control or sanitation systems. If adequate control cannot be demonstrated, the hazard would become a CCP, which has to be monitored and verified as under control.
As an example, Garelick Farms of Maine identified the following potential physical, chemical and biological hazards and determined whether these were hazards controlled by an established PP or whether they required a CCP designation. In these examples, the potential physical and chemical hazards were determined to be controlled by voluntary and mandatory PPs, respectively, and the potential biological hazard was designated as a CCP.
• Example Hazard Controlled by Voluntary Prerequisite Program. Foreign objects in dry ingredients were identified as potential physical hazards. The PP procedure outlines ingredient receiving and storage protocols, including trailer inspection and product verification. Specifically, the receiver signs off on a form for QC and labels ingredients for first-in/first-out rotation. The receiver conducts the inspection of the trailer, not only to verify that the shipment is the actual product ordered, but also to check for cleanliness of the trailer, damage to the product or to wooden pallets, and so on.
• Example Hazard Controlled by Mandatory Prerequisite Program. There are eight mandatory PPs in Phase 2 of the NCIMS pilot program, one of which is prevention of adulteration. Over-fortification of the dairy product with vitamins was identified as a potential chemical hazard under this mandatory PP, which outlines the responsibilities for operation, maintenance and calibration of vitamin pumps and the reconciliation of vitamin usage. Reconciliation is accomplished by reviewing the daily log for vitamin usage and vitamin usage is verified by periodic analysis of the product for vitamin content.
• Example Hazard Controlled by Critical Control Point. Pathogens in raw milk were identified as potential biological hazards. As such, pasteurization is the only CCP recognized in the Garelick Farms of Maine HACCP plan. Critical limits established for this CCP are those specified in the Pasteurized Milk Ordinance (PMO), although operating limits may be more stringent. The written plan specifies responsibilities for monitoring, corrective action, verification of control and records. For this example, the CCP designation would require the following specific plan:
• Monitoring: Who (pasteurizer operator); what (temperature); how (chart recorder) and frequency (continuous).
• Corrective Action: Divert and recycle product, place product on hold, evaluate, and determine disposition.
• Verification: Review recording charts (operator/QC), equipment start-up checks (operator) and quarterly equipment checks (QC/engineer ing/regulatory using PMO criteria.
• Records: Pasteurizer charts and any necessary corrective action records to note deviations from critical limits.
The pilot program also defines two different kinds of actions with regard to failures in a HACCP plan. The first, nonconformity, applies to failures to meet specified requirements of the HACCP system within the framework of a prerequisite program. For example, if the over-use of vitamin D is determined, the plant must review the cause, determine the product disposition, correct the failure and file a report in a centralized log. The second, deviation, is defined as failure to meet a critical limit at a CCP. For example, if a sub-legal pasteurization temperature is recorded, the plant must review the cause, determine the product disposition, take corrective action and file the report in a centralized log.
The Audit Component
Under the pilot program, regulatory auditors utilize a specific NCIMS HACCP audit form and conduct periodic audits of the plant’s HACCP system. One advantage of the HACCP program is that audit frequencies may be extended to every six months based on the performance of the plant. The auditor reviews any changes to the plant’s HACCP plan, such as the introduction of new products, processes or ingredients that could affect the basic premise of the HACCP plan. The auditor reviews adherence to NCIMS requirements, since some of the elements that were strictly required under the PMO remain strictly required under HACCP; for example, drug residue testing, approved milk sources, and proper product labeling requirements. The auditor should review the centralized deviation/ nonconformity logs, as well as the HACCP plan itself to ensure that specific measures are actually being implemented in the plant. The auditor will conduct a traditional plant inspection and an exit review with management personnel. Time-lines are established for corrections of any concerns noted by the auditor.
Lessons Learned
Among the lessons Garelick Farms of Maine learned by participating in the NCIMS HACCP pilot program is that the processor remains ultimately responsible for ensuring the safety of the dairy food. The company does not have to reinvent the wheel to convert to HACCP, but rather, can build off of the programs already in place. Records are vitally important, as well, and they must be user-friendly, contain all necessary information, and be verified periodically with corrective actions taken as needed.
Another lesson learned is that regulatory personnel gain greater understanding of the entire dairy manufacturing process through their review of procedures and records. HACCP promotes dialogue and compromise between regulatory and industry on procedures used to verify food safety. On a related note, a recent executive summary also emphasized these results. Specifically, in February 2001, the NCIMS HACCP Committee, which oversees the implementation of the NCIMS HACCP Pilot program for dairy plants, reported that the volunteer pilot plants and state regulators have demonstrated that dairy products manufactured under this program are as safe as under the traditional NCIMS PMO program. Based on onsite evaluations of the six pilot dairy plants conducted in late 2000 by the NCIMS HACCP Evaluation Team, the oversight committee also reported the following:
• State regulators noted that they understood the operation of the plant in more detail under the HACCP pilot and that the industry personnel were more knowledgeable and better trained regarding their public health responsibilities.
• The states have the capability to uniformly implement HACCP and provide for reciprocity between states.
• Additional time was needed to train plant personnel as well as state regulators.
• Resources required for the development, implementation, and verification of the plant’s HACCP system were greater than originally anticipated.
• The time spent on the initial audit was longer than the traditional inspection time.
• There was confusion about the relative difference between prerequisite programs, sanitation standard operating procedures, and critical control points.
• A “Hazards and Controls Guide,” as well as models of HACCP plans and prerequisite programs, should be developed.
• Auditing procedures need to be clarified and a standardization program in HACCP auditing needs to be developed.
Based on information gathered during the first fill 18 months of operation under the voluntary NCIMS HACCP system, the NCIMS HACCP Committee recommended the extension of the pilot program through 2003, and proposed expanding the program to include other states and plants that would like to participate.
Garelick Farms of Maine will continue its voluntary participation in the NCIMS HACCP pilot. As a processor not only of milk but also of juice, the plant will be implementing the mandated juice HACCP plan when the rule becomes effective in January 2002. Garelick Farms of Maine’s participation in the dairy pilot program has benefitted the company greatly in terms of being proactive in effectively preparing for successful implementation of this new rule. Participation in the NCIMS HACCP pilot program has been a positive experience reinforcing the importance of the ultimate goal of HACCP: Making product that is safe to the last drop.
Rebecca Piston is Quality Control Manager with Garelick Farms of Maine, Division of Suiza Foods, located in Bangor, ME. She was recently appointed to serve as a technical advisor to the NCIMS HACCP Committee, and has worked in dairy plant quality control for various companies since 1988. Piston holds a masters degree in food science, dairy foods microbiology from Oklahoma State University.
In May 2001, the NCIMS HACCP Committee published a new appendix in its proposals document entitled, “The Hazard Analysis Critical Control Point (HACCP) System,” which provides background information and other specifics for dairies. While the use of the HACCP system is not new to the dairy industry, the NCIMS Pilot program has shown that when properly implemented, the voluntary HACCP alternative described will provide assurance of product safety that is equivalent to that provided under the traditional system used by the dairy industry.
Appendix K defines the seven principles of HACCP as a “plan.” The HACCP Principles are:
1. Conduct a hazard analysis.
2. Determine the critical control points.
3. Establish critical limits.
4. Establish monitoring procedures.
5. Establish corrective actions.
6. Establish verification procedures.
7. Establish record-keeping and documentation procedures.
When a HACCP plan is combined with the prerequisite programs, it constitutes a HACCP “system.” The HACCP alternative described in Appendix K includes the HACCP system and other prescribed PMO criteria such as drug screening and the use of milk only from sources regulated under federal provisions. While the appendix provides more detailed information, here are a few highlights of special interest to the dairy industry:
Prerequisite Programs. PPs are the universal procedures used to control the conditions of the plant environment that contribute to the overall safety of the product. They represent the sum of programs, practices and procedures that must be applied to produce and distribute safe products in a clean, sanitary environment. The exact set of prerequisite programs will vary since their application is product- and process-specific, and these are established and managed separately from the HACCP plan. The required PPs for milk plants using the dairy HACCP alternative address:
1. Safety of the water that comes into contact with food or food contact surfaces (including steam and ice);
2. Condition and cleanliness of equipment food contact surface;
3. Prevention of cross-contamination from insanitary objects and/or practices to food products, packaging material and other food contact surfaces, including utensils, gloves, outer garments, etc., and from raw product to processed product (e.g., pasteurizer pressure differential);
4. Maintenance of hand washing, hand sanitizing, and toilet facilities;
5. Protection of food, food packaging material, and food contact surfaces from adulteration with lubricants, fuel, pesticides, cleaning compounds, sanitizing agents, condensate and other chemical, physical and biological contaminants;
6. Proper labeling, storage, and use of toxic compounds;
7. Control of employee health conditions that could result in the microbiological contamination of food, food packaging materials, and food contact surfaces; and
8. Pest exclusion from the food plant.
(Note: PPs Nos. 4 and 6 are required under the FDA Juice HACCP Regulation, 21 CFR Part 120.)
Hazard Analysis. Each milk plant, receiving station or transfer station shall develop, or have developed for it, a written hazard analysis to determine whether there are food hazards that are reasonably likely to occur for each type of milk product processed by that milk plant, receiving station or transfer station and to identify the control measures that the milk plant, receiving station or transfer station can apply to control those hazards.
A hazard that is reasonably likely to occur is one for which a prudent milk plant, receiving station or transfer station operator would establish controls because experience, illness data, scientific reports, or other information provide a basis to conclude that there is a reasonable possibility that, in the absence of these controls, the hazard will occur in the particular type of product being processed. In evaluating what food hazards are reasonably likely to occur, at a minimum, consideration should be given to the following: microbiological contamination; parasites; chemical contamination; unlawful drug and pesticide residues; natural toxins; unapproved use of food or color additives; presence of undeclared ingredients that may be allergens; and physical hazards.
Looking for quick answers on food safety topics?
Try Ask FSM, our new smart AI search tool.
Ask FSM →






