Keeping Honey Products Safe: Detecting Sulfonamides using Liquid Chromatography-Mass Spectrometry
The rules and regulations as to the content, labeling and even the very nature of components within honey is under seemingly continuous debate. With a number of contested environmental, ecological and personal health issues to consider, modern honey manufactures are faced with the challenge of meeting a host of variable regulations and ensuring consumer protection.
The treatment of honey with veterinary medicines is a particularly controversial issue and the maximum residue limits of commonly used antibiotics, such as sulfonamides, permitted within honey is commonly set at trace levels (although this can vary significantly depending on regional preferences). However, quantitative sulfonamide analysis presents substantial challenges to those working in honey manufacture, as traditional analytical techniques have difficulty achieving the selectivity, specificity and accuracy to perform accurate low-level quantitation within complex biological matrices.
Honey producers therefore require increasingly sophisticated methods to exact control over their products and meet demanding food safety criteria. Here we explore how newly developed high performance triple quadrupole liquid chromatography-mass spectrometry (LC-MS TQ) with multiple reaction monitoring (MRM) methodologies provides a fast, simple and robust technique for accurate and confident low level sulfonamide quantitation.
Detecting Sulfonamides
Sulfonamides are an important group of broad spectrum antibacterial drugs, frequently employed in the production of honey to prevent and treat potential bacterial growth, in addition to fighting disease in honey bees. Consequently, residues of sulfonamides are frequently detected in samples of commercially available honey products.
The maximum content of sulfonamide residue in honey is determined by a number of complicated personal health and environmental considerations. Sulfonamides are a common allergen and can provoke a variety of reactions in hypersensitive humans, with effects ranging from mild irritation to severe dermatological disorders. As the varied effects of allergens are dependent on the response of the individual consumer, it is technically impossible to quantify an exact concentration at which food can be classified as safe for consumption. Thus, the concentration of sulfonamide residue that will provoke a reaction in all sensitive consumers is not strictly defined and legislation pertaining to the maximum residue allowance varies considerably between regions.
Additionally, the need to control sulfonamide use goes beyond accommodating hypersensitive consumers. It has also been suggested that the use of sulfonamides in honey may contribute to development of antibiotic-resistant pathogenic bacteria. However, opinion on the level of this risk is divided, as is how best to mitigate it. This lack of clear scientific consensus regarding the safety and appropriate use of sulfonamides means that many regions set particularly low MRPLs (minimum required performance levels) for residue concentration, with the European Union limiting sulfonamide content in honey to 10 µg/kg.[1]
However low-level sulfonamide residues are notoriously difficult to analyze. The complex interactions that arise from biological matrices, such as honey, substantially obfuscate analysis as traditional techniques suffer extensive preparation times, limited selectivity and low throughput.
Multiple Reaction Monitoring Solutions
EU legislation states that the efficient control of residues is an essential component of the maintenance of a high level of consumer protection.[2] These laws define the required levels of the specificity, sensitivity and reproducibility, along with more specific rules on the requirements to assure effective quantitation. European Union Directive 2002/657/EC lists a number of potential analytical techniques of which MS [often coupled with ultra high performance LC (UHPLC)] is unarguably the most powerful technique commonly available for the identification of organic compounds within a variety of matrices.[3]
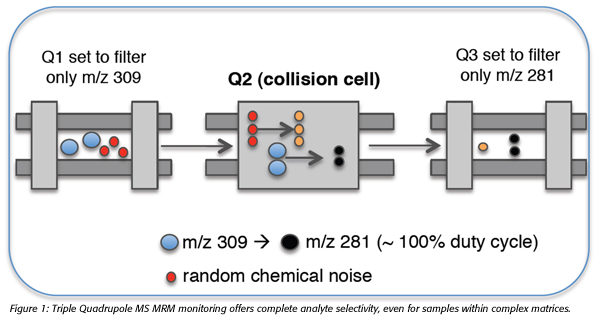 Of the range of MS techniques available, MRM with LC-MS TQ has emerged as the most effective for food analysis, favored for its high sensitivity and selectivity in complex matrices with minimal sample preparation. MRM methods of MS analysis employ a mass filtering function to enhance selectivity during analyte quantitation. Ions of interest are first preselected as ‘precursor’ ions before undergoing fragmentation within a collision cell. By then filtering only the sequence-specific fragment ions for analysis, MRM MS enables highly sensitive and easy to perform multiplexing analysis with the specificity required at low analyte levels, even within complex biological matrices (Figure 1).
Of the range of MS techniques available, MRM with LC-MS TQ has emerged as the most effective for food analysis, favored for its high sensitivity and selectivity in complex matrices with minimal sample preparation. MRM methods of MS analysis employ a mass filtering function to enhance selectivity during analyte quantitation. Ions of interest are first preselected as ‘precursor’ ions before undergoing fragmentation within a collision cell. By then filtering only the sequence-specific fragment ions for analysis, MRM MS enables highly sensitive and easy to perform multiplexing analysis with the specificity required at low analyte levels, even within complex biological matrices (Figure 1).
Certain MRM MS systems have now evolved to provide higher levels of specificity and selectivity for food testing through innovative advances in instrument design. For example, the EVOQ Elite from Bruker features a unique active exhaust concept that improves the sensitivity of the MS TQ by eliminating gas recirculation within the instrument, so improving noise reduction for increased sensitivity. Additionally, developments in practical features, such as performing ion transfer through an orifice rather than capillary interface, focus on increasing both ion transport efficiency and operational ease of use by avoiding disruptive routine blockages.
New work by Bruker has explored how MRM LC-MS/MS can be employed specifically for the analysis of sulfonamides within honey samples. A new methodology has been developed that ensures accuracy in analysis, substantially decreases routine analysis time and adheres to European Union Directive 2002/657/EC.
Case Study: Using MRM LC-MS/MS to Analyze Sulfonamides in Honey
To illustrate the capacity of this new analytical methodology, an experiment was carried out into the simultaneous determination of sulfonamide residues in honey using Advance UHPLC and the EVOQ Elite LC-MS TQ.[4]
 Nine commonly used sulfonamide antibiotics were analyzed using the new method: sulfadimethoxin (SMM), silfadiazin (SD), sulfadosin (SDM), sulfamethazine (SM1), sulfadimidine (SDN), sulfamerazine (SM1), sulfathiazole (ST), suldamethoxypyridazin (SMP) and sulfamethizole (SMZ). The method’s repeatability was studied on three different days by spiking blank honey matrices with levels of 0.1, 1 and 10 ppb sulfonamides. Figure 2 illustrates the Advance UHPLC and EVOQ Elite instrumentation set up respectively.
Nine commonly used sulfonamide antibiotics were analyzed using the new method: sulfadimethoxin (SMM), silfadiazin (SD), sulfadosin (SDM), sulfamethazine (SM1), sulfadimidine (SDN), sulfamerazine (SM1), sulfathiazole (ST), suldamethoxypyridazin (SMP) and sulfamethizole (SMZ). The method’s repeatability was studied on three different days by spiking blank honey matrices with levels of 0.1, 1 and 10 ppb sulfonamides. Figure 2 illustrates the Advance UHPLC and EVOQ Elite instrumentation set up respectively.
Sample Preparation
Commercially available honey (2.5 g) was acidified with 2 mL of 1% formic acid in water and vortexed for 2 minutes, followed by the addition of 8 mL LC-MS/MS-grade acetonitrile and 10 minutes of ultrasonic extraction. One gram of powder from BR73763 Kit was added and the sample thoroughly shaken for 30 seconds. This was mixed and vortexed for 15 minutes to achieved complete solubilization before undergoing centrifugation for 3 minutes at 3000 rpm.
The top 4 mL of the organic layer was transferred to another centrifuge tube and evaporated to dryness at 45 oC under nitrogen. The extract was reconstituted with 0.5 mL initial mobile phase [water:MeOH (80:20)] and filtered through a 0.22-µm nylon fiber filter directly into an HPLC vial and injected into the system. This simple sample preparation procedure allows the detection of all targeted compounds, drastically reducing routine preparation time from hours to little over 30 minutes.
Results and Discussion
 Figure 3 shows representative chromatograms of all nine sulfonamides investigated at limit of quantification (LOQ) concentration level of 0.1 ppb in honey with peak-to-peak S/N ratio. The S/N ratios for all nine sulfonamides within the honey matrix at 0.1 ppb is well above 10. Results obtained for all nine sulfonamides under study at LOQ (0.1 ng/g)-level concentration were found to be well within the acceptable range of accuracy and precision.
Figure 3 shows representative chromatograms of all nine sulfonamides investigated at limit of quantification (LOQ) concentration level of 0.1 ppb in honey with peak-to-peak S/N ratio. The S/N ratios for all nine sulfonamides within the honey matrix at 0.1 ppb is well above 10. Results obtained for all nine sulfonamides under study at LOQ (0.1 ng/g)-level concentration were found to be well within the acceptable range of accuracy and precision.
 By comparison the blank honey matrix is, as to be expected, clean of targeted sulfonamides. Figure 4 shows the overlaid chromatogram of all nine sulfonamides at LOQ level and the blank honey matrix.
By comparison the blank honey matrix is, as to be expected, clean of targeted sulfonamides. Figure 4 shows the overlaid chromatogram of all nine sulfonamides at LOQ level and the blank honey matrix.
To provide greater stringency and more precaution against false positives, the European Union approach to MRM testing requires four matching MS ion identification points to ensure identification of organic residue and contaminants in samples of animal origin. For forbidden Group A substances, such as sulfonamides, a minimum of four identification points are required to assure detection of interest.
 This requirement for four identification points (IPs) is satisfied through the acquisition of two MRM transitions where 1.0 is assigned for the ‘precursor ion’ and 1.5 IPs are assigned for each target analyte. The ion ratio of a pair of MRM transitions acquired for each target analyte must match to those of the certified reference material within specified tolerance. These were found to be consistent for all the analyte samples taken for this study. Figure 5 illustrates this for sulfamethazine.
This requirement for four identification points (IPs) is satisfied through the acquisition of two MRM transitions where 1.0 is assigned for the ‘precursor ion’ and 1.5 IPs are assigned for each target analyte. The ion ratio of a pair of MRM transitions acquired for each target analyte must match to those of the certified reference material within specified tolerance. These were found to be consistent for all the analyte samples taken for this study. Figure 5 illustrates this for sulfamethazine.
 Calibration curves for all nine sulfonamides reveal excellent linear response with R2 values in excess of 0.996 (Figure 6). The method was also found to be extremely repeatable (intra-day and inter-day) with relative standard deviations below 12%, again satisfying an important specification of the EU directive.
Calibration curves for all nine sulfonamides reveal excellent linear response with R2 values in excess of 0.996 (Figure 6). The method was also found to be extremely repeatable (intra-day and inter-day) with relative standard deviations below 12%, again satisfying an important specification of the EU directive.
When performed with the EVOQ Elite LC-MS/MS TQ system, the MRM-based sulfonamide analysis demonstrated excellent detection limits and repeatability for all nine sulfonamides under study. Additionally, the use of Advance UHPLC and simplified experimental set up reduced both sample preparation and chromatographic run time, offering the potential for higher throughput routine analysis. The validation results revealed that the method can be used for the screening and quantification of medicinal sulfonamides commonly used in honey production, to the level required by Europe and other nations.
Conclusion
The demands made of the food industry to perform safety testing with the confidence needed to ensure product quality and consumer safety has stimulated development of smarter analytical techniques and testing methodologies. Today, MRM LC-MS/MS is the gold standard for food safety testing and methodologies—such as those developed by Bruker using the EVOQ Elite for honey analysis—are providing the accuracy, speed and ease of use required for efficient food screening.
Joe Analceto and Gouri Satpathy are with Bruker Chemical and Applied Markets. For more information, please visit www.bruker-daltonics.com.
References
1. European Union Decision 2002/657/EC: Commission Decision of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results (Text with EEA relevance) [notified under document number C(2002) 3044].
2. ec.europa.eu/food/food/chemicalsafety/residues/lab_analysis_en.htm.
3. Lehotay S.J. and K. Mastovska. 2008. Identification and confirmation of chemical residues in food by chromatography-mass spectrometry and other techniques. Trends Anal Chem 27(11):1070–1090.
4. Satpathy, G. Quantitative LC-MS/MS analysis of sulphonamides in honey using advanced UHPLC-EVOQ Elite LC-MS/MS system. Bruker Daltonics application note #1824322.
Looking for quick answers on food safety topics?
Try Ask FSM, our new smart AI search tool.
Ask FSM →





