FDA
Restaurants should not sell or serve recalled mushrooms
Read More
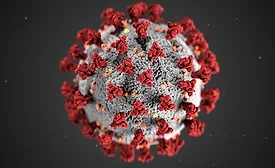
COVID-19 coverage
Pandemic prompts temporary policy changes
Facilities can use FDA guidance to comply with FSMA during the coronavirus crisis
August 17, 2020
Regulatory Watch
FDA outlines steps to help prevent recurring Salmonella outbreaks linked to papayas
Federal regulators issue second letter to papaya growers and others in the industry
August 11, 2020
Never miss the latest news and trends driving the food safety industry
Newsletters | Website | eMagazine
JOIN TODAY!Copyright ©2026. All Rights Reserved BNP Media, Inc. and BNP Media II, LLC.
Design, CMS, Hosting & Web Development :: ePublishing